The subspine organization of actin fibers regulates the structure and plasticity of dendritic spines.
Synapse function and plasticity depend on the physical structure of dendritic spines as determined by the actin cytoskeleton. We have investigated the organization of filamentous (F-) actin within individual spines on CA1 pyramidal neurons in slices of rat hippocampus. Using two-photon photoactivation of green fluorescent protein fused to β-actin, we found that a dynamic pool of F-actin at the tip of the spine quickly treadmilled to generate an expansive force (Fig. A,B). The size of a stable F-actin pool at the base of the spine depended on spine volume (Fig. C,D). Repeated two-photon uncaging of glutamate formed a third pool of F-actin and enlarged the spine (Fig. E,F). Creation of this "enlargement pool" required calmodulin. The spine often released the enlargement pool into the dendritic shaft, but the pool had to be physically confined by a spine neck for the enlargement to be long-lasting (Fig. G,H). Ca2+/calmodulin-dependent protein kinase II regulated this confinement. Thus, we have revealed that spines have a mechanical nature that is regulated by the three pools of F-actin.
Program member
Haruo Kasai (Laboratory of Structural Physiology, Center for Disease Biology and Integrative Medicine, Graduate School of Medicine)
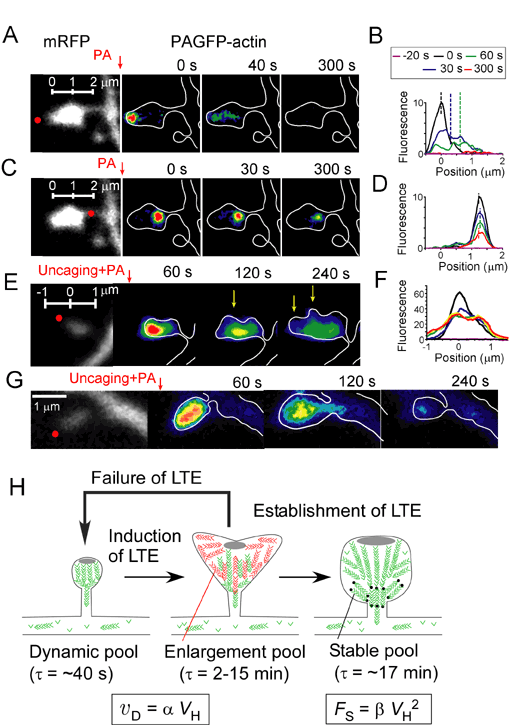
A,C,E,G, The cytosol of spine was stained with mRFP (gray), and actin was labeled with PAGFP (pseudo-color code).B,D,F, Fluorescence profile of PAGFP. H, LTE stands for long-term enlargement of spine-head volume. VH, vD and Fs represent the spine-head volume, velocity of actin polymerization and size of the stable pool of actin fibers.
